Moderna seeks emergency use authorization for COVID-19 booster shots for all adults
Moderna chairman on COVID boosters, new therapeutic
Moderna Chairman Noubar Afeyan argues there could be a ‘continues need for boosting’ every year.
Moderna filed a request with the Food and Drug Administration on Wednesday for authorization of its COVID-19 vaccine booster shots for all Americans aged 18 and older.
The company is seeking an emergency use authorization for the 50-microgram dose, which is half of the 100-microgram administered when patients receive their first two doses.
ORLANDO, FLORIDA, UNITED STATES – 2021/09/25: Dr. Salma Elfaki holds a vial of Moderna COVID-19 vaccine at a Moderna clinical trial for adolescents being conducted by Accel Research Sites with Nona Pediatric Center in Orlando. (Photo by Paul Hennessy ( Paul Hennessy/SOPA Images/LightRocket via Getty Images / Getty Images) In October, FDA officials authorized Moderna’s booster shot for adults aged 65 or older, as well as for adults between 18 and 64 who are at high risk for severe COVID-19 or have "frequent institutional or occupational exposure" to the virus. The boosters are administered at least six months after the patient completed their vaccination regimen. PFIZER/BIONTECH COVID-19 BOOSTER 95.6% EFFECTIVE AGAINST DISEASE: STUDY This June 14, 2021, file photo shows a Moderna COVID-19 vaccine vial that is being administered for flight attendants of Japan Airlines ar Haneda Airport in Tokyo. (AP Photo/Eugene Hoshiko / AP Newsroom) Earlier this month, Pfizer-BioNTech filed a request with the FDA seeking an emergency use authorization for its own COVID-19 booster shot for adults 18 and older. Seattle Mariners fans walk past signs for the Johnson & Johnson and Moderna COVID-19 vaccines, Tuesday, May 4, 2021, during a clinic held at T-Mobile Park before a baseball game between the Mariners and the Baltimore Orioles in Seattle. (AP P (AP Photo/Ted S. Warren) CLICK HERE TO READ MORE ON FOX BUSINESS Both the FDA and the Centers for Disease Control and Prevention must approve the boosters for specific age groups before they can be administered. The FDA is expected to sign off on Pfizer’s booster shot on Thursday, the New York Times reported. Source: Read Full Article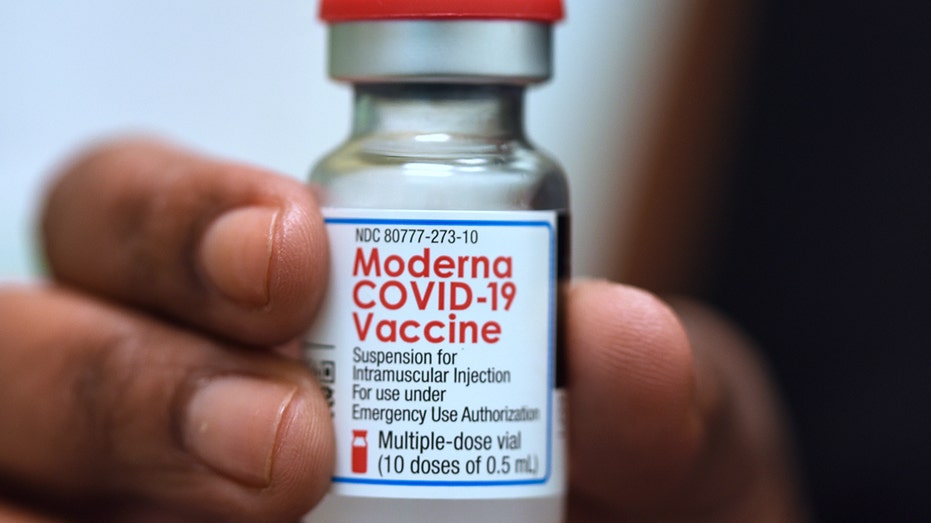
Ticker
Security
Last
Change
Change %
MRNA
MODERNA, INC.
242.23
+7.95
+3.39%

